Two data events in the same week can do remarkable damage to a stock, especially when the narrative around that stock was already stretched. First came the bone mineral density scare: a Cantor Fitzgerald analyst flagging a 4% BMD decline from Phase I data published eight months earlier, data that Amgen's own management had previously acknowledged and dismissed. Then came the Phase II MariTide readout itself, up to 20% average weight loss at 52 weeks, a genuinely impressive clinical result that the Street had nonetheless priced for something bigger. AMGN fell nearly 20% from its 2024 highs. The market delivered its verdict swiftly.
But markets often get the sentiment right and the analysis wrong. At $260–$280, you are buying a company generating $8.5 billion in quarterly revenue, growing 23% year-over-year, with 33% net margins, 12 consecutive years of dividend increases, and a forward P/E of 14.3x. The GLP-1 option value is free. The question is not whether MariTide will be perfect. The question is whether the bar for "good enough to compete" in a market where Eli Lilly and Novo Nordisk together command $1.18 trillion in combined market cap is actually as high as the current share price implies.
This note walks through the core business Amgen has quietly been building, the Phase II data without the noise of the stock price reaction, the structural dynamics of a market large enough to absorb multiple winners, and the scenarios that define this position's risk-reward. The thesis is not that MariTide will win. The thesis is that Amgen wins even in most of the losing scenarios, and the upside of even partial success has not been captured in the current price.
The Core Machine: What 23% Growth Actually Looks Like
The noise around MariTide has obscured something important: Amgen's underlying commercial franchise is performing at a level few of its peers can match. In Q3 2024, total revenues hit $8.5 billion, up 23% year-over-year. Ten drugs posted double-digit sales growth in that single quarter. This is not a company running on fumes while it waits for a pipeline win.
The growth engine has several distinct components. Repatha, the PCSK9 inhibitor for cardiovascular risk, grew 25% year-over-year in Q3 driven by 46% volume expansion, partially offset by lower net prices under managed care agreements. Evenity, the bone-building drug for osteoporosis, grew 39%. Blincyto, the leukemia treatment and the clearest example of Amgen's BiTE antibody platform delivering, grew 28%. Tezspire, the anti-TSLP biologic developed with AstraZeneca for severe asthma, grew 76% year-over-year to $234 million in a single quarter. These are not rounding errors in a financial model. They represent genuine volume growth across multiple therapeutic areas.
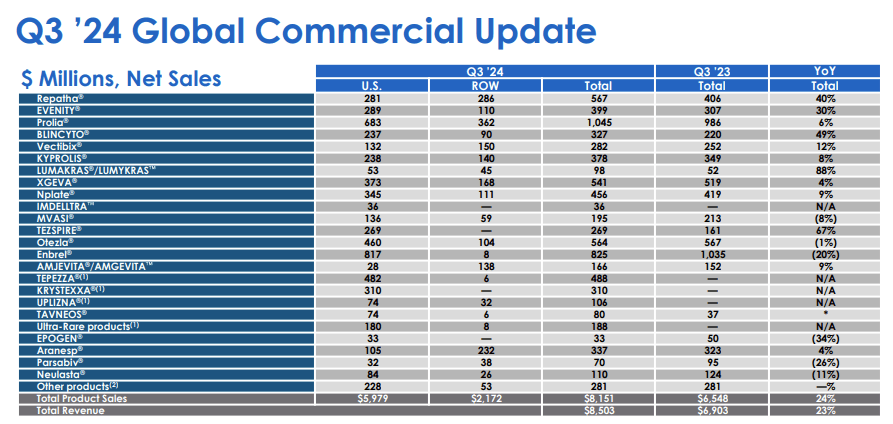
The Horizon Therapeutics acquisition, completed in October 2023 for approximately $28 billion, is also beginning to show its strategic value. Horizon products added $1.1 billion in revenue for Q3 alone. Tepezza, the thyroid eye disease drug, grew 8% year-over-year with a Japan launch targeting early 2025. Krystexxa for uncontrolled gout generated $294 million. Tavneos, which Amgen acquired separately through ChemoCentryx, grew 137% year-over-year, and the number of patients prescribed it has more than doubled since the acquisition. The integration has gone well, and what looked to some analysts like an expensive empire-building exercise is proving its commercial logic.
Looking at the forward picture, consensus estimates project revenues growing from $33.18 billion in 2024 to $40.05 billion by 2030. That 20.7% revenue growth is expected to translate into 43.6% EPS expansion, from $19.57 per share to $28.10 per share, because of operating leverage and Amgen's sustained share buyback program. The share count has already been reduced 25.7% between 2017 and 2023, from 720.6 million shares to 535.9 million. That trajectory, if continued, means EPS growth will almost certainly outpace the consensus model.
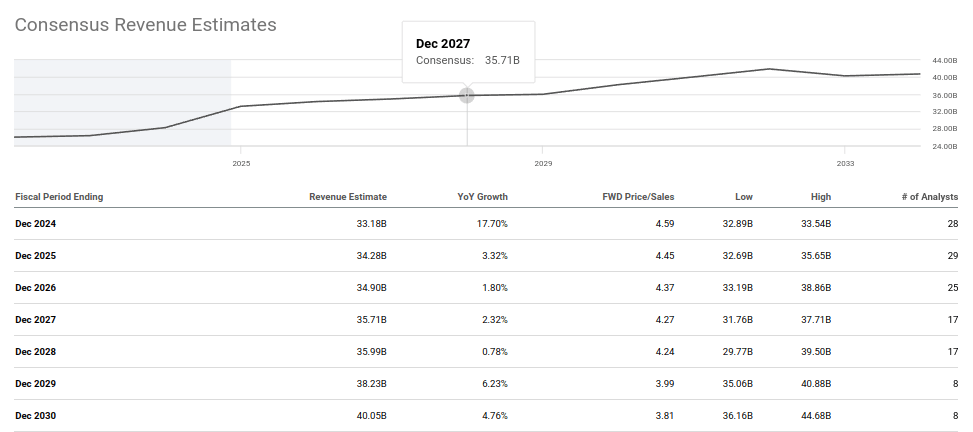
The critical observation here is that these consensus revenue estimates were built largely before MariTide was a serious commercial factor. They model organic growth from the existing portfolio. That means the $33B-to-$40B trajectory represents a floor, not a ceiling. A successful MariTide adds on top. An unsuccessful MariTide leaves the model largely unchanged. At 14.3x forward earnings, even the floor looks undervalued.
Reading MariTide Phase II Without the Stock Price
The standard advice in clinical-stage investing is to analyze data before looking at the stock price reaction. In practice, almost nobody does this. The price is there, it moved violently, and it shapes how every subsequent data point is processed. Let me try to do the exercise properly.
MariTide (maridebart cafraglutide, AMG 133) is a novel bispecific molecule that works via two distinct mechanisms: it activates the GLP-1 receptor, the same target as semaglutide and tirzepatide, and it simultaneously inhibits the GIP receptor (GIPR). The genetic rationale for the GIPR inhibition is that people with naturally reduced GIPR signaling show protection against obesity and related metabolic conditions. This dual mechanism is what Amgen has consistently presented as MariTide's differentiation: not just a "me-too" GLP-1 agonist, but a fundamentally different intervention in the obesity biology.
The Phase II study enrolled 592 participants in total, with 465 in the obesity cohort. Patients were divided across dose arms of 140mg, 280mg, and 420mg, administered monthly via subcutaneous injection, against a placebo arm. The key efficacy result: up to 20% average weight loss at 52 weeks, without a plateau.
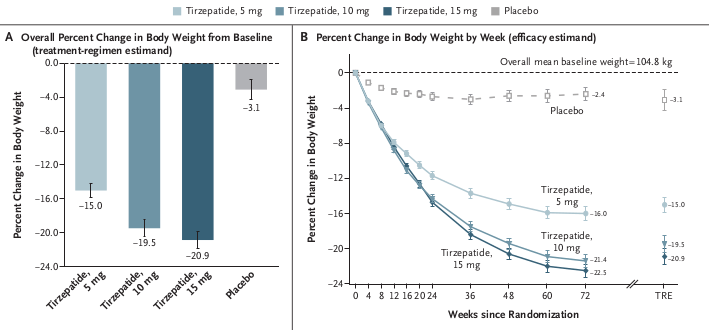
The "disappointment" narrative requires a benchmark. The most relevant comparison is tirzepatide (Zepbound/Mounjaro), Eli Lilly's current standard-bearer, whose Phase III SURMOUNT-1 trial reported 20.9% weight loss at 72 weeks for the highest dose. MariTide delivered 20% at 52 weeks. That is 20 fewer weeks of treatment. An apples-to-apples comparison, extrapolating the MariTide curve to 72 weeks without a plateau, would put MariTide in a directly competitive weight-loss range with tirzepatide, somewhere in the 22% to 23% territory.
A one-year study stopping before the curve plateaus is a structural advantage for the Phase III narrative, not a weakness. Ninety percent of Phase II patients elected to continue for a second year. That is not the behavior of patients experiencing unacceptable side effects or unsatisfying weight loss. That is a retention signal.
This trajectory was not a surprise to Amgen's team. Phase I repeated-dosing data had already shown a consistent downward weight curve with no sign of flattening at the final measured time-point. That early kinetic profile is what informed the Phase II design decision to run the trial to 52 weeks rather than a shorter window, and it is precisely why the absence of a plateau in Phase II carries such weight: the same pattern was visible from the very first human dosing studies.
On safety, three specific concerns dominated the market reaction and are worth addressing individually.
Bone mineral density: The Cantor Fitzgerald analyst raised this based on Phase I data published in February showing a 4% BMD decline at the highest dose. The Phase II data definitively resolved this: no adverse impacts on bone mineral density were observed in the 52-week trial. The concern was a false alarm based on preliminary data.
GI side effects: The discontinuation rate in dose-escalation arms was approximately 11% for any adverse event, with less than 8% for GI-specific events. This compares unfavorably to tirzepatide's Phase III, where the highest discontinuation rate for adverse events was 7.1%. However, context matters. In the MariTide arms employing an escalation strategy starting from 70mg, nausea rates fell below 50% and vomiting below 20% by day 43. More importantly, 90% of patients completing the year elected to continue. Amgen explicitly states that GI events were "predominantly mild to moderate, transient, and primarily associated with the first dose" and that "subsequent doses were extremely well tolerated."
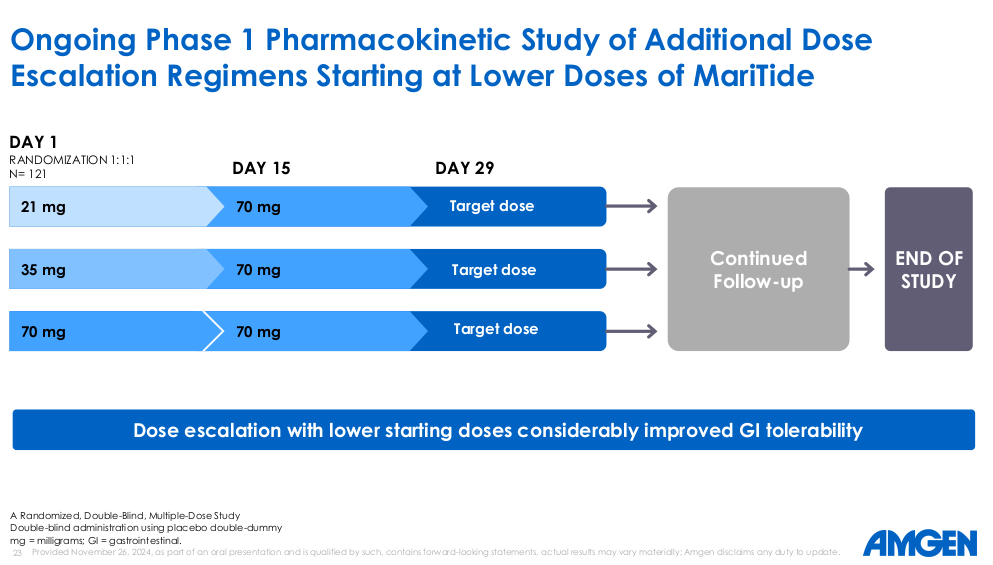
Discontinuation rate uncertainty: The market reacted negatively to limited data granularity around which patients discontinued and at which dose. The intriguing observation is that the 280mg cohort showed comparable or slightly superior efficacy to the 420mg cohort. This raises the possibility that the FDA-approved dose in Phase III may not be 420mg at all, which would naturally improve the safety profile while preserving the efficacy. The dose escalation strategies being explored are compelling and suggest Amgen's team is thinking clearly about the commercial rollout.
The conclusion from a cold reading of the Phase II data: this is not a drug in trouble. This is a drug that showed competitive weight loss efficacy at half the duration of the benchmark, maintained a 90% continuation rate, resolved its bone density concern, and has a clearly identifiable tolerability strategy for Phase III. One analyst, who covered MariTide Phase I closely, raised his estimated probability of MariTide becoming a commercial drug from 60% to approximately 80% following Phase II. That upward revision after the data that "disappointed" the market is worth pausing on.
"We are fully confident that MariTide's differentiated profile can contribute meaningfully to addressing the epidemic of obesity, the burden of type 2 diabetes, and other serious obesity-related conditions."
Jay Bradner, Executive VP of R&D and Chief Scientific Officer, Amgen - Phase II Update, November 26, 2024The Obesity Race: Why the Market Is Large Enough for a Third Player
The framing of GLP-1 as a winner-take-all competition is a narrative convenience, not a medical reality. Consider how the precedents actually played out in comparable market categories. Statins did not produce one winner. Antihypertensives did not produce one winner. Biologics for rheumatoid arthritis, where Humira, Enbrel, Remicade, and Xeljanz all coexisted at multi-billion-dollar scale, is perhaps the most instructive parallel. The obesity market at $500 billion-plus in addressable potential by the end of the decade is not a niche. It is an expansion of a metabolic disease category that will require multiple treatment approaches, patient populations with different tolerability profiles, and healthcare systems with different reimbursement frameworks.
Where specifically does MariTide compete? The dosing frequency argument is compelling and underappreciated. Both semaglutide and tirzepatide are administered weekly. GLP-1 users have high discontinuation rates in the real world, often 30% to 50% within the first year of treatment, particularly because the weekly injection routine combined with persistent GI side effects creates patient fatigue. Monthly dosing, or even quarterly dosing (which Amgen is exploring in a Phase II sub-study), is not just a convenience feature. It is a fundamental change in the patient experience that could drive materially higher adherence rates in a market where adherence is currently the biggest commercial risk for existing products.
The manufacturing angle is also worth taking seriously. Lilly and Novo Nordisk have both faced significant supply shortages of Zepbound and Ozempic/Wegovy. These shortages are structural: tirzepatide and semaglutide are peptide-based medicines that require specialized manufacturing capacity with long lead times. MariTide, by contrast, is built on a monoclonal antibody backbone that fits directly into Amgen's existing manufacturing network. Amgen's CFO Peter Griffith flagged this explicitly on the Q3 call: "yield is really important, and we've got a really strong history in that." The R&D budget is being stretched to prepare: Q3 R&D spend was up 35% versus the prior year quarter, and up over 25% for full-year 2024. The Phase III MARITIME program has been launched, and Amgen is investing at scale to be ready.
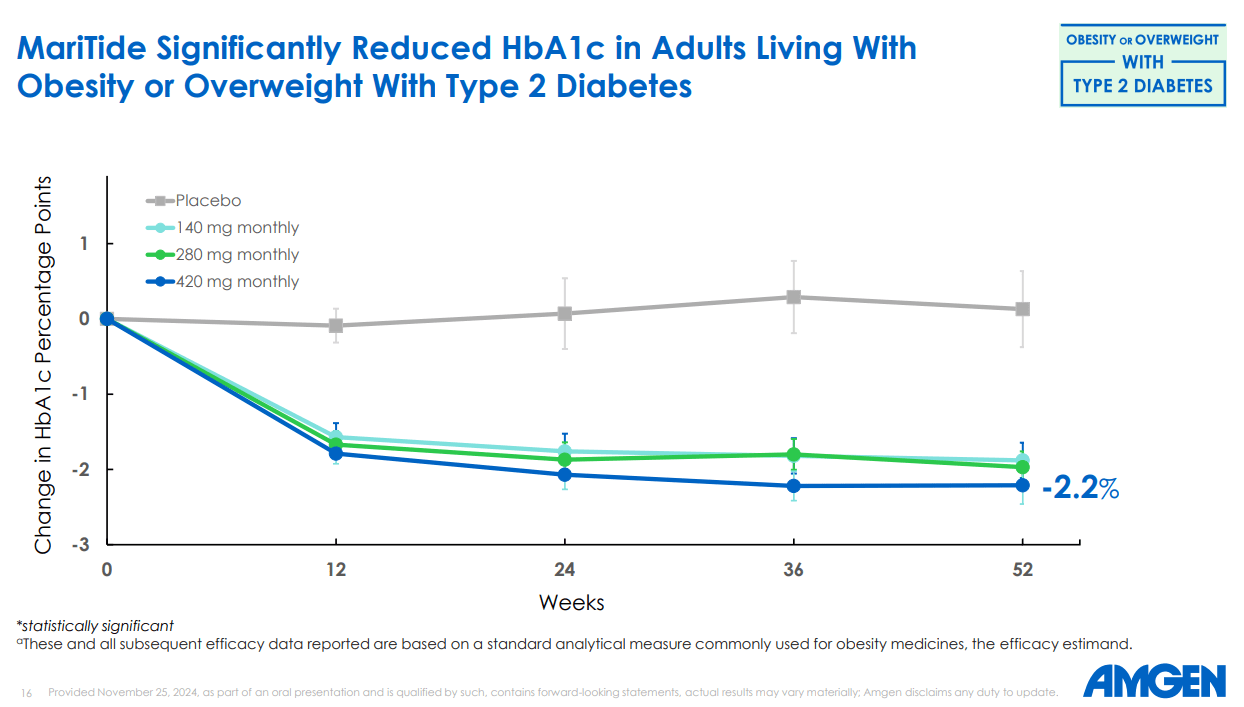
Then there is the type 2 diabetes angle, which most market commentary has underweighted. In the T2D cohort of the Phase II study, MariTide delivered 17% weight loss at 52 weeks alongside a 2.2 percentage point reduction in HbA1c, clinically meaningful improvements in systolic blood pressure, triglycerides, and hs-CRP. The HbA1c reduction is within the range of what CagriSema, Novo Nordisk's next-generation candidate, achieved in its Phase II. And MariTide has a monthly dosing advantage there too. As Susan Sweeney, Amgen's Executive VP of Obesity, put it on the Phase II call: MariTide has "the potential to be the first and only monthly treatment option" for type 2 diabetes, "a long-standing goal in the field."

Morningstar estimated MariTide's addressable market at $8 billion annually by 2033, with a 60% approval probability before the Phase II data. With Phase II having confirmed competitive efficacy and resolved the bone density concern, that probability estimate has moved upward. Zooming out to the broader obesity treatment landscape - estimated to reach $500 billion annually at full market maturity - even a 5% share would represent approximately $25 billion in annual revenue within a decade, against Amgen's current total annual revenue of $33 billion. Partial success in GLP-1 changes the company's growth trajectory irreversibly.
What the Market Priced In vs. What the Data Actually Show
The $260–$280 price implies a specific interpretation of the available evidence. Here is the most honest way to map what drove the sell-off versus what the data actually say when you read them without the stock price in your peripheral vision.
- BMD concern from 8-month-old Phase I data, flagged the week before Phase II readout
- 20% weight loss at 52 weeks fell short of Street expectations near 25%+
- 11% total adverse event discontinuation rate, above tirzepatide's 7.1% Phase III benchmark
- Limited transparency on safety data at initial announcement, creating information vacuum
- CagriSema and retatrutide (Lilly) potentially showing 24-27% weight loss in coming years
- Perceived "crowded" GLP-1 market with two deeply entrenched leaders
- RFK Jr. named to lead HHS, raising general uncertainty about drug pricing policy
- BMD concern definitively resolved in Phase II: no adverse impact observed in 52-week trial
- Weight loss at 52 weeks without plateau implies 22-23% by 72 weeks on current trajectory - this is an analytical extrapolation, not a trial result. It assumes linear continuation of the efficacy curve with no plateau effect; biological weight-loss curves typically do slow over time, so the actual 72-week outcome could be lower. The comparison with competitor data is therefore directional rather than definitive, but it illustrates the plausible competitive range if MariTide's trajectory holds.
- 90% patient retention after one year: strongest real-world adherence signal in the class
- Dose escalation strategy demonstrably reduces GI events to competitive range
- T2D cohort shows clinically meaningful HbA1c reduction of 2.2pp, potential first monthly therapy
- Monthly dosing solves the adherence problem that weekly products struggle with at scale
- Manufacturing platform requires no new capacity: existing Amgen network sufficient for global scale
The priced-in narrative says: MariTide is too weak on efficacy, too toxic on safety, arriving too late to a race already won by Lilly and Novo. The data narrative says: competitive efficacy with a differentiated dosing profile, improving tolerability, and a structural manufacturing advantage in a market with a 30-50% real-world attrition problem for existing weekly therapies. Both views are interpretations of the same data. The question is which one is worth $270 per share versus $350 per share.
The Bear Case: Honest Engagement with the Risks
Any credible analysis of Amgen at this juncture has to spend serious time with the patent cliff. This is not a hypothetical risk. It is a scheduled event. Denosumab biosimilars (biosimilars to Prolia and Xgeva, combined 2023 sales of $6.2 billion) are slated to launch in May 2025 following a court-approved settlement with Sandoz. Enbrel, the TNF inhibitor for rheumatoid arthritis and psoriasis, has been experiencing structural net price declines for years and generated $3.7 billion in 2023 versus meaningfully higher figures earlier in the decade. Otezla, acquired from Bristol-Myers Squibb in 2019 for $13.4 billion as a condition of the BMS-Celgene merger, saw a 4% revenue decline in 2023 as competition intensified.
The counter-argument is that the consensus model already reflects these headwinds. The $33B-to-$40B revenue trajectory through 2030 accounts for biosimilar erosion in Prolia/Xgeva and continued Enbrel pressure. What has offset the declines is the same portfolio growth described in Section 1: Repatha, Evenity, Blincyto, Tezspire, Kyprolis, and the Horizon rare disease franchise growing rapidly enough to more than compensate. Amgen's design philosophy, articulated by CEO Robert Bradway, is explicitly to build a portfolio designed "to deliver growth through volume increases" rather than price increases, making it structurally more resilient to pricing headwinds from legislation like the Inflation Reduction Act.
On MariTide specifically, the bear case is not that it is a bad drug. The bear case is that the GI tolerability profile, despite dose escalation improvements, may limit commercial adoption relative to Wegovy or Zepbound in the real world, where patients are less closely monitored than in clinical trials. If 20-25% of patients cannot tolerate the titration even with the optimized protocol, MariTide may end up as a niche product for patients who have failed first-line GLP-1 therapy rather than a primary market entrant.
The debt load is also worth noting. Amgen's debt-to-equity ratio stands at 8.02, elevated largely because of the Horizon acquisition. The interest burden constrains financial flexibility during the Phase III investment cycle, when R&D spending will increase materially. Free cash flow is robust (Amgen generated $9.0 billion in operating cash flow in 2023), but capital allocation will be under pressure simultaneously from dividend increases, share buybacks, debt service, and MARITIME trial costs.
None of these concerns are fatal. They do, however, set the parameters of the bear scenario: a world in which MariTide fails Phase III or achieves only marginal market penetration, the patent cliff bites harder than modeled, and the stock drifts toward its 2023 trough valuation near 12x forward earnings, which would imply a price around $230–$240 on 2025 EPS estimates. That is approximately 15% downside from the entry zone. It is a defined, manageable risk against a base case that suggests 25-35% upside and a bull case that approaches 50% upside.
What a $190 Price Target Spread Actually Signals
Ten analysts have current ratings on Amgen. Their price targets range from $215 to $405, a $190 spread on a $270 stock. That spread is not a sign of poor analysis. It is a calibrated reflection of exactly the kind of binary optionality that creates asymmetric opportunity for investors who form an independent view.
Brian Skorney at Baird, the lone Underperform at $215, is essentially making the following argument: the patent cliff is worse than modeled, MariTide will not achieve sufficient commercial differentiation, and the stock should trade closer to its 2023 trough multiple. This is a coherent view and one worth understanding thoroughly. The bulls at Cantor Fitzgerald ($405, Overweight), Jefferies ($380, Buy), TD Cowen ($383, Buy), and Bernstein ($380, Outperform) are pricing in a world where the existing portfolio continues growing and MariTide captures meaningful obesity market share. The neutral camp at Citigroup, UBS, Truist, and Barclays is essentially saying: the risk-reward from here is balanced and they want more clarity on Phase III design before committing to a directional view.
The street consensus price target of $349 represents approximately 28-34% upside from the entry zone of $260–$280. That consensus price target is, importantly, not a bull-case target. It is the average of 11 views that range from deeply cautious to aggressively optimistic. It is the point at which the market collectively stops fighting the valuation.
One additional signal worth registering: Michael Yee at Jefferies labeled the bone density concern a "non-issue" when it first surfaced, maintained his $380 Buy rating through both data events, and has not wavered. Yee is one of the more closely followed biotech analysts at a major bank. When the analyst who raised the loudest initial warning (Brayer at Cantor Fitzgerald) maintains a $405 Overweight through the Phase II readout while adding the drug's bone safety credentials were confirmed, that tells you something about the underlying conviction on the bull side of this trade.
The Howard Chang Signal: Reading Between the Lines of a Senior Hire
On November 20, 2024, exactly one week before the MariTide Phase II readout, Amgen announced the appointment of Howard Chang as Chief Scientific Officer, effective December 16. The timing looked almost deliberate: a company preparing to make the case that it is serious about long-term scientific leadership, not just riding the GLP-1 wave.
Chang is not a conventional pharma executive hire. He is a Stanford professor who has spent two decades researching chromatin biology, RNA, and epigenetics, and who has co-founded multiple biotechnology companies. He holds undergraduate and medical degrees from Harvard, a PhD in biology from MIT, and has been a faculty member at Stanford since 2004. He is the kind of hire that a company makes when it intends to compete on discovery science for the next decade, not just optimize the commercial performance of an existing pipeline.
"Howard is one of our generation's foremost physician-scientists, with expertise in human genetics and a profound ability to distill complex disease biology into clarified targets."
Jay Bradner, EVP R&D, Amgen - announcing Howard Chang's appointment as CSO, November 20, 2024The hiring context matters. Leerink Partners recently ranked Amgen among the top five large-cap biopharma companies exposed to patent expirations for blockbuster drugs in the 2025–2030 period. That exposure is real. What Chang represents is Amgen's long-term answer to that exposure: a research engine capable of generating the next generation of first-in-class medicines across rare disease, oncology, cardiometabolic, and inflammation, the same four pillars CEO Bradway articulates consistently.
The Blincyto BiTE platform is already delivering on that ambition. Imdelltra (tarlatamab), the DLL3-targeting BiTE therapy, received FDA accelerated approval in May 2024 for small cell lung cancer, demonstrating 40% response rates and 14.3-month median overall survival in a disease where under 3% of patients survive five years. A new CSO with Chang's background signals that this platform approach will be extended, not merely maintained. For investors with a multi-year horizon, this hire is a signal about where Amgen will be in 2030, not just 2025.
Catalyst Roadmap: What to Watch and When
The timeline for this position has clear milestones on both the positive and risk sides. Understanding the sequencing is important for sizing and managing the trade.
Outcome Assessment: Three Scenarios
This is a position with genuine optionality. The three scenarios below are designed to be honest: they reflect downside cases that are plausible, not strawmen constructed to make the base look inevitable.
Phase III confirms superior efficacy of CagriSema and retatrutide. MariTide's GI profile remains unfavorable despite dose escalation, driving commercial prescriber preference toward existing weekly therapies. Patent cliff bites harder than modeled; Enbrel and Prolia declines accelerate. Stock reverts to 12x forward EPS, near 2023 trough multiples.
The base business still generates substantial FCF and dividends continue. Floor is supported by a conservative $19+ EPS base.
$230–$250 ~15% downside from entry midpointMariTide is approved in 2027-2028. Monthly dosing drives materially better real-world adherence versus weekly competitors. Captures 5-8% of obesity market and first-mover position in monthly T2D treatment. Core franchise grows as modeled: $40B revenue by 2030, EPS approaching $28.
Stock re-rates from 14.3x to 17-18x forward earnings as analysts gain confidence in the combined commercial and pipeline narrative. Consistent dividend growth continues.
$340–$370 ~28-37% upside from entry midpointPhase III data shows MariTide achieving 22-24% weight loss at 72 weeks with a substantially improved tolerability profile under optimized escalation. T2D approval follows. Quarterly maintenance dosing studied and validated. Amgen captures 10%+ of the obesity market. New CSO Chang accelerates discovery pipeline; Blincyto BiTE platform delivers additional oncology approvals.
Amgen is reframed as the biotech that made the obesity market a three-horse race. Market cap closes the gap toward Novo/Lilly peers. Revenue trajectory revised significantly above consensus.
$390–$430 ~46-60% upside from entry midpointFinal Thought: When the Market Gives You the Option Value for Free
Rare moments emerge in biotech investing when a single noisy data event sends a high-quality business to a valuation that makes no sense independent of the binary outcome. Amgen at $260–$280 is one of those moments. The arguments from five independent research frameworks converge on the same conclusion through different analytical lenses.
The quantitative frame: 14.3x forward earnings for a company with 33% net margins, 42% return on equity, 23% revenue growth, and 12 consecutive dividend increases. That multiple is historically cheap for Amgen, which spent most of the past five years between 16x and 20x. The trough was 12x in the depths of the 2023 post-Horizon acquisition skepticism. You are buying near the trough.
The clinical frame: Phase II delivered competitive weight loss efficacy without plateau, resolved the bone density concern, and showed a 90% patient retention rate. The GI tolerability issue has a clear mechanistic solution in dose escalation. Management is moving forward with Phase III with conviction rather than caution. The probability of MariTide becoming a commercial drug, based on analysts who have followed the program since Phase I, moved upward, not downward, after the Phase II data.
The competitive structure frame: the obesity market will not be won by one drug. Weekly injectables with persistent GI side effects and supply constraints create a structural opening for a monthly alternative. The analogy is not insulin, where price competition commoditized the category. The analogy is antihypertensives, where multiple mechanisms, dosing frequencies, and tolerability profiles coexist at multi-billion-dollar scale simultaneously.
The execution frame: Robert Bradway opened the Q3 earnings call by addressing MariTide directly, before discussing any financial results. Peter Griffith stated that MariTide "is right at the front" of R&D investment priorities. Susan Sweeney launched the MARITIME Phase III program. Howard Chang joined as CSO with a mandate to accelerate discovery. These are not the actions of a management team losing confidence. They are the actions of a team that has seen the Phase II data in full and is moving at speed.

The asymmetry frame: the downside scenario prices AMGN at $230–$250, roughly 15% below the entry midpoint, and still implies a dividend-paying, cash-generative business. The base scenario, which requires only MariTide capturing a modest slice of the obesity market with its monthly dosing advantage, puts the stock at $340–$370. The bull scenario, which requires genuine Phase III outperformance, reaches $390–$430. The risk-reward ratio of approximately 1:2.5 in the base case and 1:3.5 in the bull case reflects what asymmetric option value looks like when the market has overreacted to uncertainty.
Morningstar's pre-Phase II estimate put MariTide at 60% odds of approval. Several analysts revised that figure upward after Phase II data. The market's price action implied the opposite: a sharp downgrade of MariTide's commercial viability. This kind of divergence between probability revision and price action is the fundamental raw material of investment opportunity. It requires forming an independent view of the data and holding it against the emotional narrative of a 15% single-day move. This note is an attempt to do exactly that.
"With a strong, balanced portfolio of in-market products and a rapidly advancing pipeline of innovative medicines, we are confident in our ability to deliver attractive long-term growth."
Robert A. Bradway, Chairman and CEO, Amgen - 2024 Annual Letter to ShareholdersAt $260–$280, the core franchise alone is arguably worth $290–$320 on conservatively discounted cash flows. The GLP-1 option is priced at approximately zero. The Phase III program is fully funded and underway. The management team is experienced, aligned, and moving with conviction. And a new Chief Scientific Officer with world-class credentials in human genetics just joined to build the pipeline after MariTide. Whatever the obesity drug's eventual commercial fate, the discount at which the full business currently trades does not reflect the quality of what you are buying.
MariTide vs. the Competition: Where It Fits
For context on where MariTide sits within the evolving GLP-1 landscape, this comparison uses data available at the time of the Phase II readout in November 2024. Phase III outcomes for next-generation candidates are projected.
| Drug | Company | Mechanism | Dosing | Best Efficacy Reported | Stage | Differentiator |
|---|---|---|---|---|---|---|
| Semaglutide (Wegovy) | Novo Nordisk | GLP-1 agonist | Weekly SC | ~15% at 68 weeks | Approved | First-in-class, global brand |
| Tirzepatide (Zepbound) | Eli Lilly | GLP-1/GIP dual | Weekly SC | ~21% at 72 weeks | Approved | Best-in-class efficacy currently |
| MariTide (AMG 133) | Amgen | GLP-1 agonist / GIPR inhibitor | Monthly SC | ~20% at 52 weeks (no plateau) | Phase III | Monthly dosing, manufacturing scale |
| CagriSema | Novo Nordisk | GLP-1/amylin dual | Weekly SC | ~24% projected (Phase III) | Phase III | Higher efficacy ceiling |
| Retatrutide | Eli Lilly | GLP-1/GIP/glucagon triple | Weekly SC | ~24-27% projected (Phase III) | Phase III | Highest projected efficacy class |
The landscape confirms the thesis: MariTide does not need to be the best drug. It needs to be good enough to earn prescriber preference among patients who prioritize monthly over weekly dosing, patients with supply access issues on weekly therapies, and the T2D population where monthly treatment represents a category-first outcome. All three are achievable targets.
AMGN - Scenario Trade Setup
Entry zone reflects the post-Phase II sell-off trough, November 2024. The risk reference level aligns with 2023 trough multiples applied to 2025 EPS estimates: approximately 12x $19.50 forward earnings. The target zone corresponds to the base scenario 17-18x multiple on 2026 forward EPS of $21–$22, plus partial MariTide option value. Position sizing: given binary optionality from MARITIME Phase III, a moderate position with capacity to add on any further weakness below $260 is appropriate. Dividend yield at entry: approximately 3.4%, providing partial downside cushion. Key watch: Phase III design disclosure in Q1 2025 and pace of Prolia/Xgeva biosimilar erosion following May 2025 market entry.
Educational and Analytical Content Only. This analysis is produced by the PolyMarkets Investment Research team for informational and educational purposes. It reflects the research team's analytical perspective based on publicly available information as of the publication date (November 27, 2024) and does not constitute personalized investment advice, a solicitation to buy or sell any security, or a guarantee of future outcomes. Clinical trial data, pipeline timelines, and financial estimates are subject to material revision. Biotech investments carry elevated risk including binary trial outcomes, regulatory decisions, and pricing pressures. Readers should conduct their own due diligence and consult a qualified financial professional before making investment decisions. Past performance of any security or sector discussed is not indicative of future results.